Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette
Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette
- Developed for both nasal and nasopharyngeal testing
- Registered at Austrian Federal Office for Safety and Healthcare as in-vitro diagnostic devices for self-testing
- Registered at The Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, BfArM)
- Registered at the FDA under Emergency Use Authorization (EUA)
- Boxes contains 20 test kits, but also smaller volumes will be available in the future
- Comes with either single packed vials with buffer solution or one large bottle of buffer solution
- CE marked
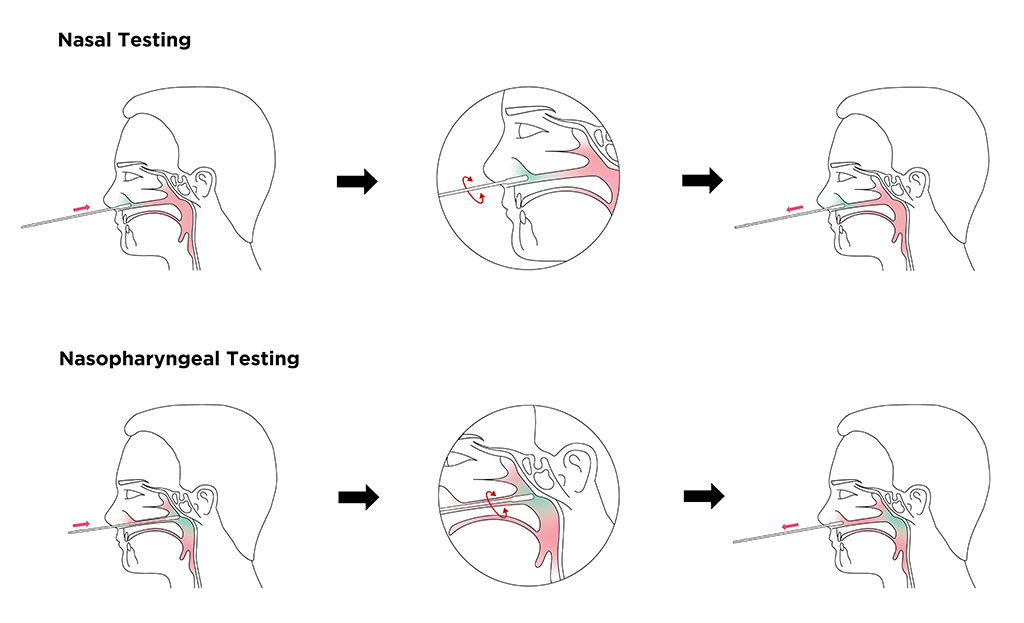
Nasal Testing
- 92.5% Sensitivity (97.7% at Ct <32)
- 99.2% Specificity
- Result in 15-20 minutes
Clinical evaluation
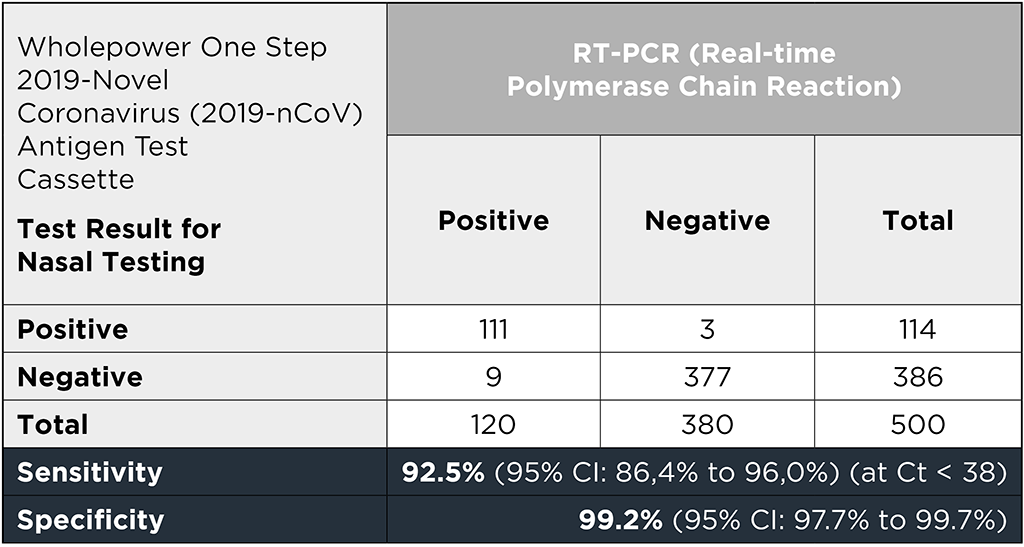
The clinical evaluation of the diagnostic performance of Wholepower’s One Step2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette is based on nasal samples from 120 RT-PCR Covid-19 positive and 380 RT-PCR Covid-19 negative individuals. All individuals were tested within seven days of symptom onset. The 120 RT-PCR-positive and the 380 RT-PCR-negative samples were collected as nasopharyngeal swaps, as this detection method is considered the golden standard in the diagnosis of COVID-19 (1).
Wholepower’s One Step 2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette showed a sensitivity of 92.5% (95% CI: 86.4% to 96.0%) for Ct values <38 and a specificity of 99.2% (95% CI: 97.7% for 99.7%). For RT-PCR-positive nasopharyngeal samples with a Ct value <32, the sensitivity of Wholepower's One Step 2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette was 97.7% (2).
- Wang X et al. Comparison of nasopharyngeal and oropharyngeal swabs for SARS-CoV-2detection in 353 patients received tests with both specimens simultaneously. Int JInfect Dis 2020;94:107-109.
- Romero-Gómez MP et al. Ct value is not enough to discriminate patients harbouringinfective virus. J Infect. 2020;26:S0163-4453(20)30720-9.
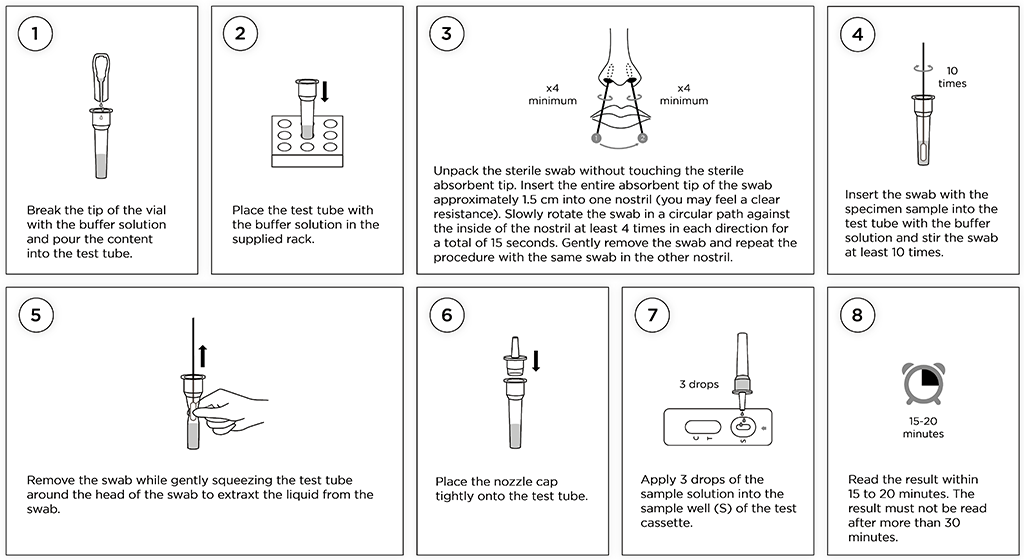
Test Procedure – Step by Step for nasal testing using vials with buffer solution
Open the box and identify the folded tube rack. Assemble the tube rack and place it on a flat surface. Now open the sealed aluminium pouch by tearing at the small notch and place the test cassette next to the rack. Then follow the instructions to the right - step by step.
Nasopharyngeal Testing
- 96.4% Sensitivity (98,8% at Ct < 32)
- 99.2% Specificity
- Results in 15-20 minutes
Clinical evaluation
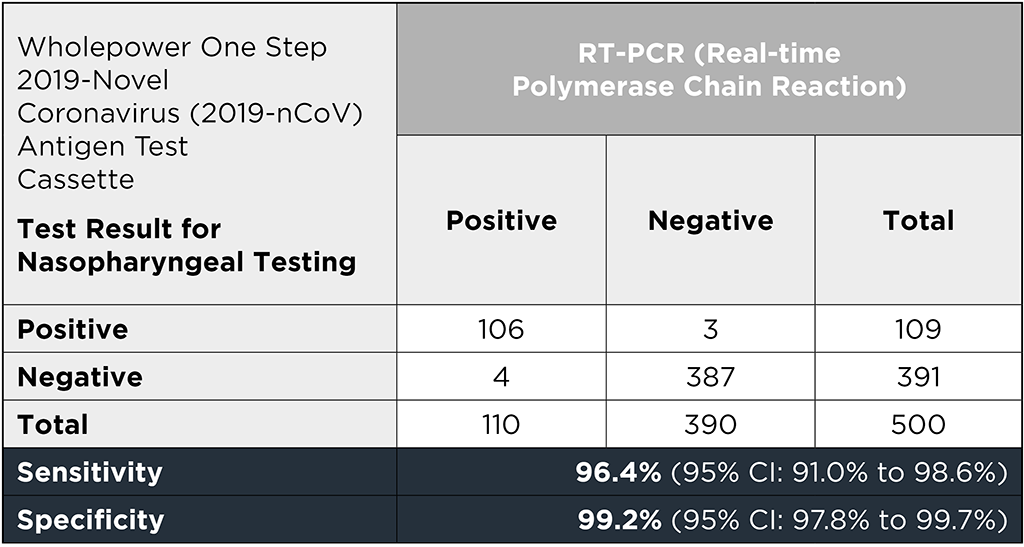
For the evaluation of the diagnostic performance of the Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) Rapid Antigen Test, RT-PCR Covid-19 positive samples from 110 individuals and RT-PCR Covid-19 negative samples from 390 individuals were evaluated in this study.
As the table shows, the test has a sensitivity of 96.4% (95% CI: 91.0% to 98.6%) and a specificity of 99.2% (95% CI: 97.8% to 99.7%).
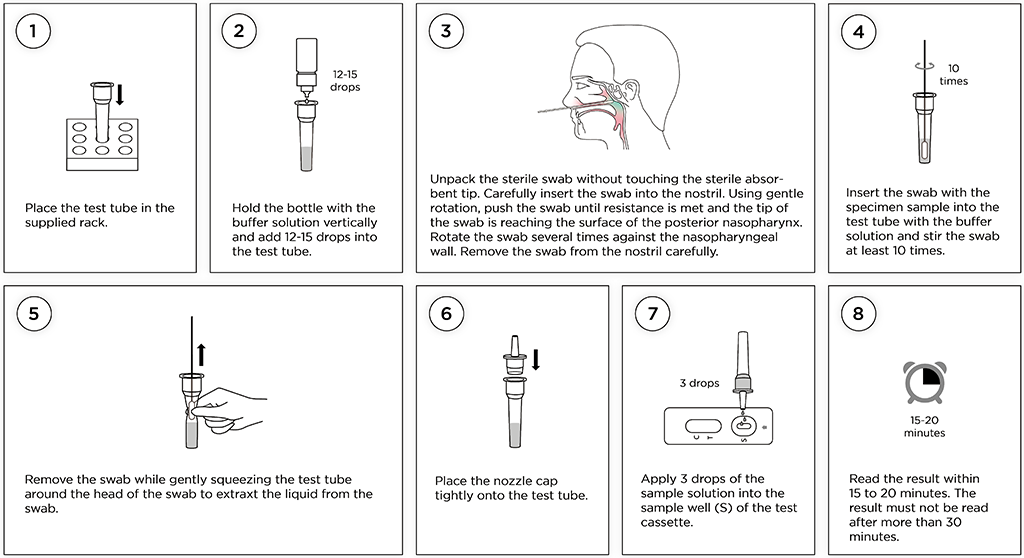
Test Procedure – Step by Step for nasopharyngeal testing using a bottle with buffer solution
Open the box and identify the folded tube rack. Assemble the tube rack and place it on a flat surface. Now open the sealed aluminium pouch by tearing at the small notch and place the test cassette next to the rack. Then follow the instructions to the right - step by step.
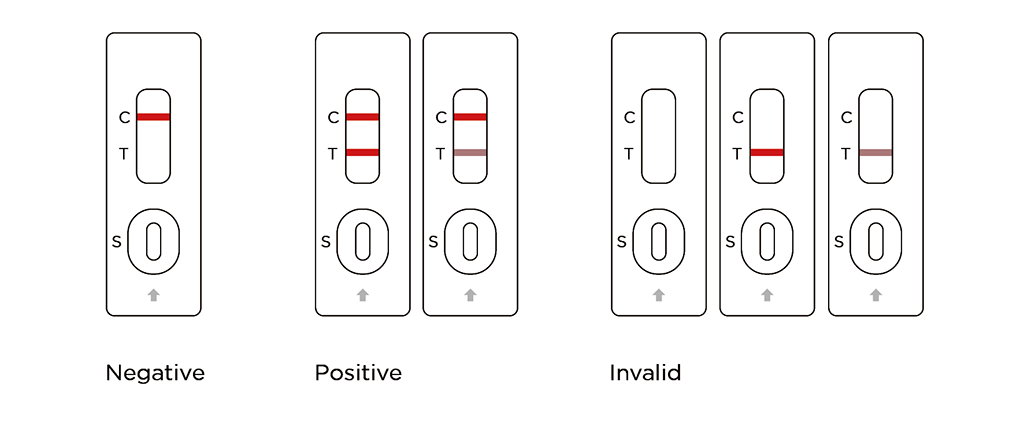
Reading the results
Negative:
Only one colored line appears in the control region (C).
Positive:
A colored line appears in the test region (T) and in the control region (C).
Invalid:
No visible line at all, or there is a visible line only in the test region (T) and not in the control region (C). Repeat with a new test kit.
Product Information
Wholepower’s One Step 2019-Novel Coronavirus (2019-nCoV) Antigen Test Cassette is a colloidal gold immunoassay for the qualitative determination of the SARS-CoV-2 nucleocapsid protein antigen (small fragments of the corona virus) in nasal or nasopharyngeal swabs, using the double antibody sandwich method.
Anti-SARS-CoV-2 nucleocapsid protein antibody (Test Line (T)) and goat anti-mouse IgG antibody (Control Line (C)) are used as the solid of the assay. The anti-SARS-CoV-2 nucleocapsid protein antibody can bind with the SARS-CoV-2 nucleocapsid protein antigen in the specimen. During the test procedures, the SARS-CoV-2 nucleocapsid protein antigen in the specimen will bind with the colloidal gold-anti-SARS-CoV-2 nucleocapsid protein antibody specifically. Because of capillary and chromatographic effects of the Nitrocellulose membrane, the complexes migrate along the membrane to the anti-SARS-CoV-2 nucleocapsid protein antibody line (T), form Abα-AG-Abβ-AU complexes and remain captured in the T line. As a result, a red coloured line develops in T and the result is positive. If there is no SARS-CoV-2 nucleocapsid protein antigen in the specimen, there is no red coloured line in the Test zone, indicating a negative result. No matter if there’s SARS-CoV-2 nucleocapsid protein antigen in the specimen, when the complexes migrate along the Control Zone, a red coloured line must develop in the C zone. If no red coloured line develops in the C zone the test is invalid.