
Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) IgG/IgM Rapid Antibody Test
Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) IgG/IgM
Rapid Antibody Test
- 89.56% Sensitivity
- 99.81% Specificity
- Stability tested
- Transport stability tested
- Results in <15 minutes
- CE marked
Clinical evaluation
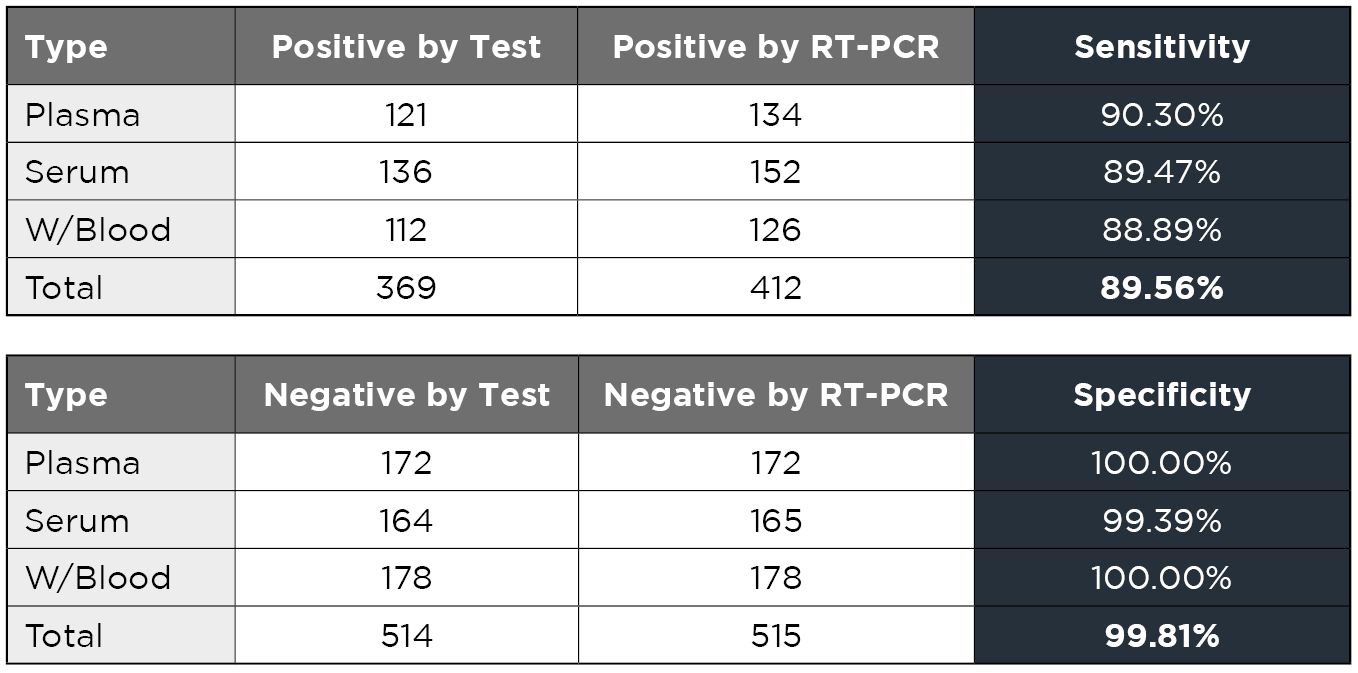
For the evaluation of the diagnostic performance of the Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) IgG/IgM Rapid Antibody Test, RT-PCR Covid-19 positive samples from 412 individuals and RT-PCR Covid-19 negative samples from 515 individuals were evaluated in this study.
As the table shows, the test has a sensitivity of 89.56% and a specificity of 99.81%.
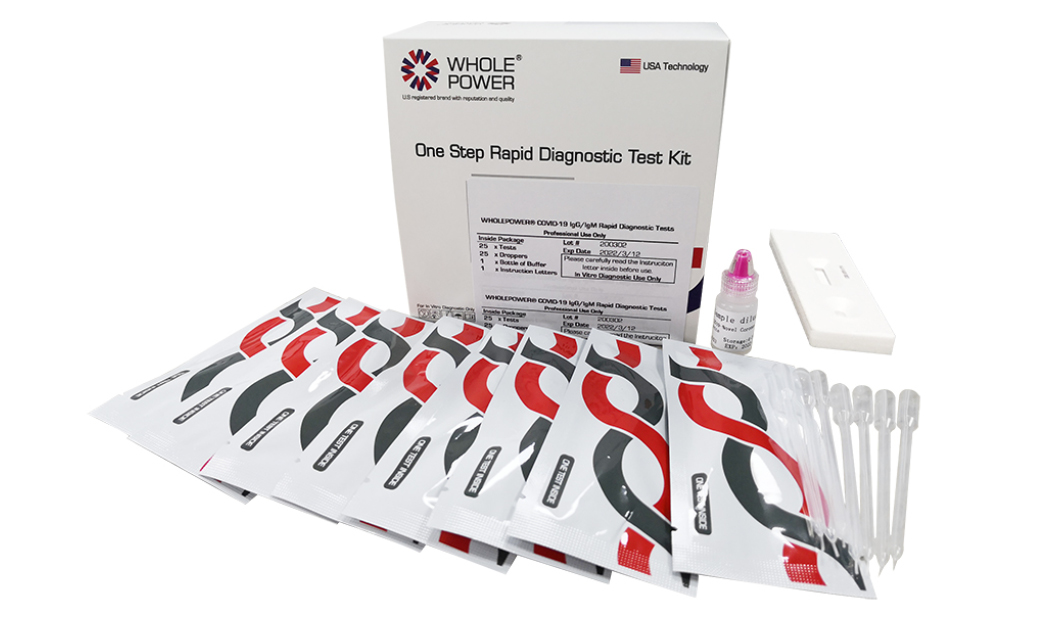
Product Information
The Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) IgG/IgM Rapid Antibody Test Cassette is a colloidal gold enhanced capture immunoassay for the qualitative determination of COVID-19 IgG and IgM antibody in human serum, plasma and whole blood samples.
The product is pre-embedded with recombined COVID-19 antigen and chicken IgY antibody on the nitrocellulose membrane, and coated with anti-human IgM, anti-human IgG and goat anti-chicken IgY antibody respectively on the G line, M line and C line on the nitrocellulose membrane.
When a positive sample is tested, the COVID-19 IgM and/or IgG antibody combines with the colloidal gold labeled COVID-19 antigen to form a complex.
Under the action of chromatography, the complex flows along the membrane, when it passes G line and/or M line it combines with the anti-human IgM and/or IgG to form a colloidal gold complex to show color, and the gold colloidal labeled chicken IgY antibody combines with goat anti-chicken IgY antibody at C line to show color. Negative samples only show color on C line.

Test Procedure
Specimen collection and preparation
- The Wholepower One Step 2019-Novel Coronavirus (2019-nCoV) IgG/IgM Rapid Antibody Test is suitable for serum, plasma or whole blood samples. The common used anticoagulants (heparin, EDTA or sodium citrate) have no effect on the results of this kit.
- Samples should be collected according to routine clinical methods and avoid hemolysis.
- If serum or plasma specimens are to be retested in 5 days, they should be refrigerated at 2°C~8°C. Storage at -20°C should not exceed 3 months. For long time storage, samples should be -70°C cryopreservation and avoid repeated freezing and thawing (no more than 3 times).
- Whole blood samples can be stored at 2°C~8°C kept in refrigerator for 3 days with no cryopreservation.
- Restore the sample to room temperature before test.
- Obvious hemolysis, lipemia and jaundice samples should not be used.
- Transportation of the samples should be sealed ice cup with ice or sealed foam box with ice.
Sample Preparation
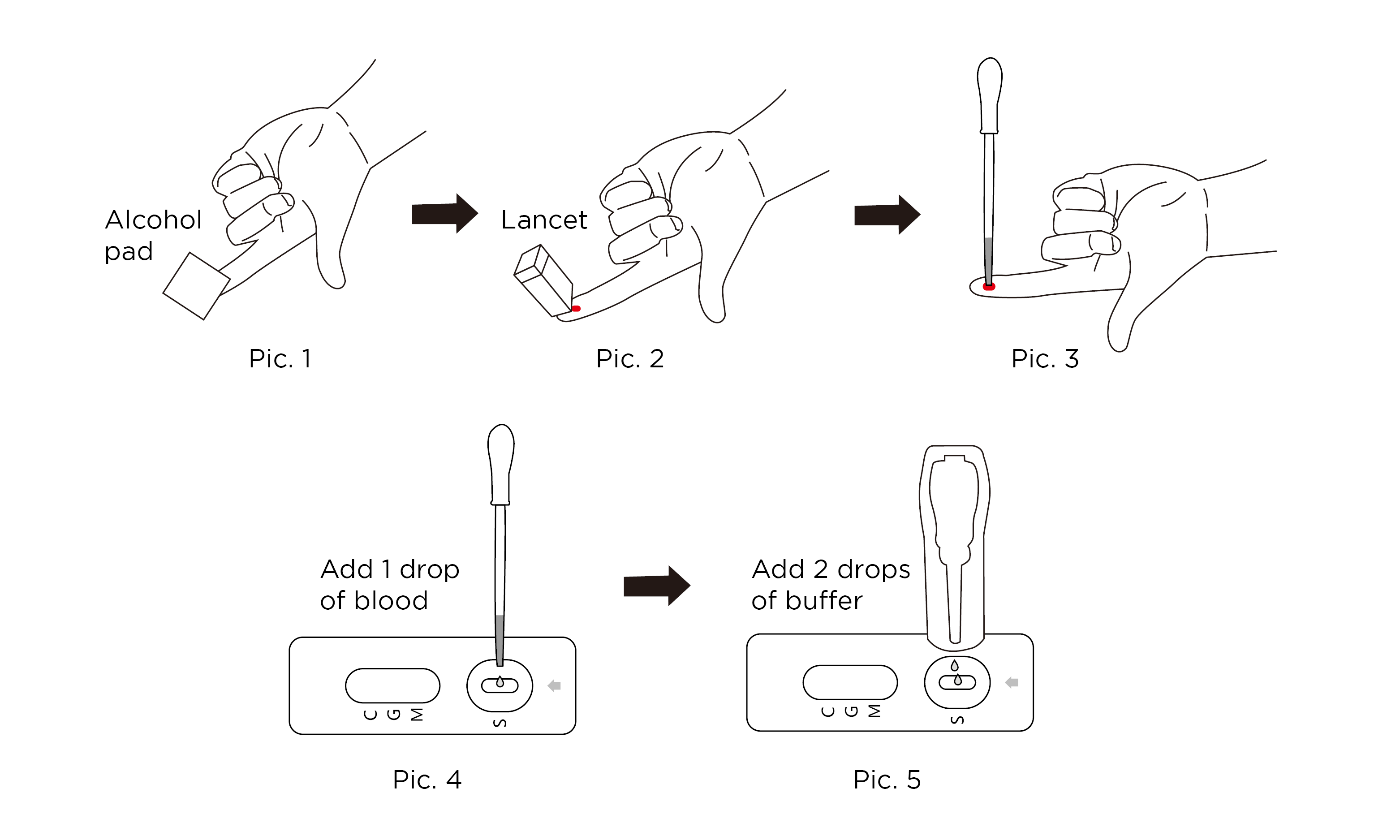
- Remove a Test from the foil pouch by tearing at the notch and place it on a flat surface.
Disinfect finger tip with alcohol pad (Pic. 1). - Pierce the finger tip with the lancet (Pic. 2).
- Use one of the disposable pipettes to collect blood from the finger end (Pic. 3).
- Holding the sample pipette vertically, add 1 drop of blood to the sample well (Pic. 4).
- Then add 2 drops of buffer to the sample well (Pic. 5).
- Read the result in 10-15 minutes. Do not read results after more than 20 minutes.
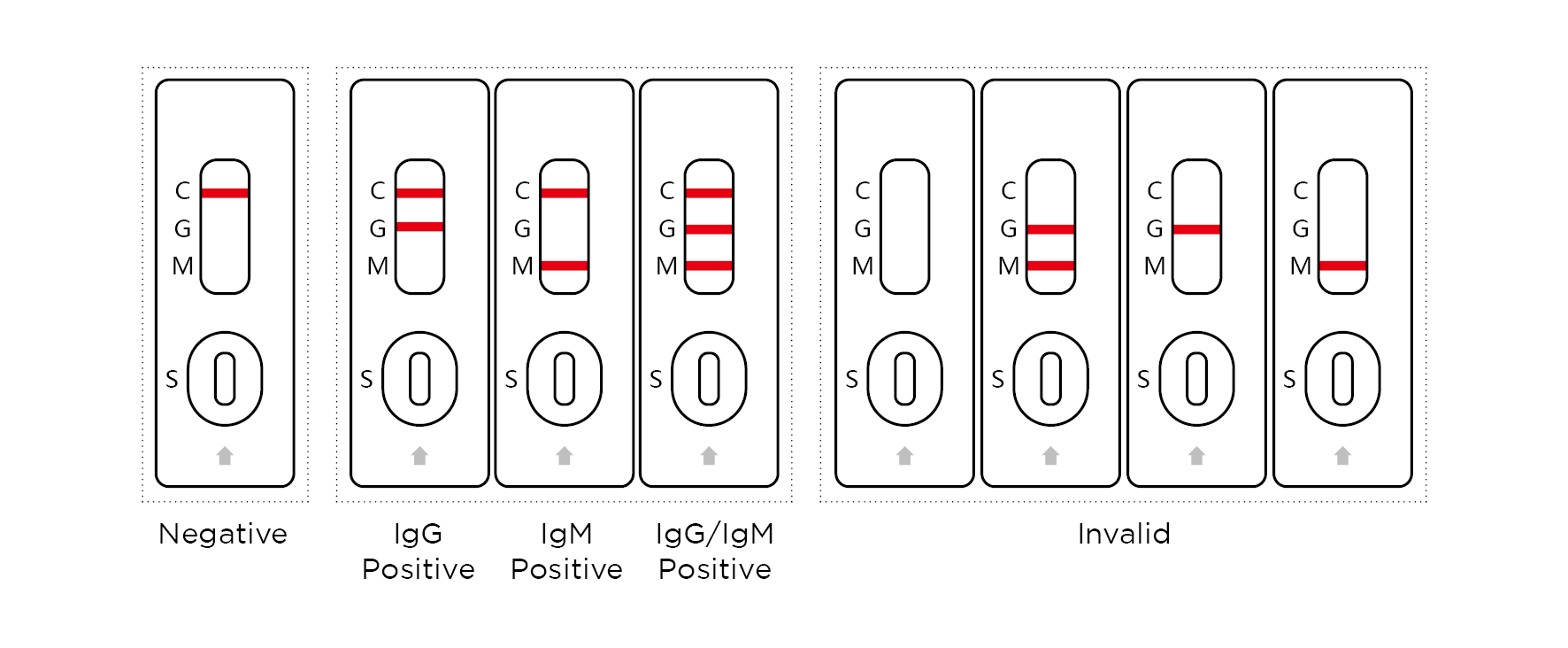
Reading the results
Negative:
Only one colored line appears in the control region (C).
Positive:
COVID-19 IgG Positive: Colored lines appear in both IgG test region (G) and control region (C).
COVID-19 IgM Positive: Colored lines appear in both IgM test region (M) and control region (C).
COVID-19 IgG/IgM Positive: Colored lines appear in test regions (G and M) and control region (C).
Invalid:
No visible line at all, or there is a visible line only in the test region and not in the control region. Repeat with a new test kit.



